Research Highlights
Whole-tissue stretch regulates epithelial cell fate decisions
Adrien Hallou, Ben Simons and colleagues at the Cambridge Stem Cell Institute and at the Gurdon Institute show that physiological mechanical forces during postnatal development control cell fate decisions in the oesophagus epithelium and triggers the transition to adult tissue homeostasis.
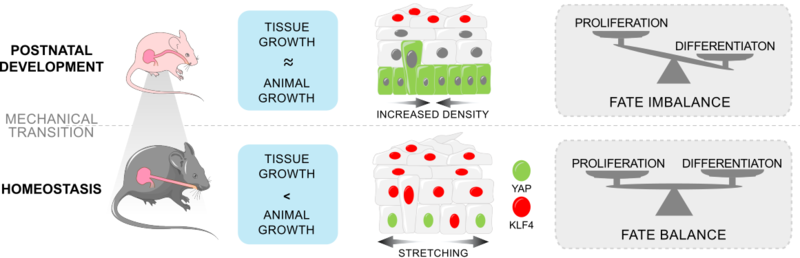
Schematic of the suggested model for the biomechanical switch controlling cell fate decisions during oesophagus epithelium postnatal development.
Epithelial cells rapidly adapt their behaviour in response to increasing tissue demands. However, the processes that finely control these cell decisions remain largely unknown. The postnatal period covering the transition between early tissue expansion and the establishment of adult homeostasis provides a convenient model with which to explore this question. In this study, we demonstrate that the onset of homeostasis in the epithelium of the mouse oesophagus is guided by the progressive build-up of mechanical strain at the organ level. Single-cell RNA sequencing and whole-organ stretching experiments revealed that the mechanical stress experienced by the growing oesophagus triggers the emergence of a bright Krüppel-like factor 4 (KLF4) committed basal population, which balances cell proliferation and marks the transition towards homeostasis in a yes-associated protein (YAP)-dependent manner. Our results point to a simple mechanism whereby mechanical changes experienced at the whole-tissue level are integrated with those sensed at the cellular level to control epithelial cell fate.
A biomechanical switch regulates the transition towards homeostasis in oesophageal epithelium Nature Cell Biology 23 511 (2021)