Sinisa Vukovic

Dr Sinisa Vukovic
Postdoc in Dr Huggins's group
Office: 516 Mott Bld
Phone: +44(0)1223 3 37238
Email: sv375 @ cam.ac.uk
TCM Group, Cavendish Laboratory
19 JJ Thomson Avenue,
Cambridge, CB3 0HE UK.
Research
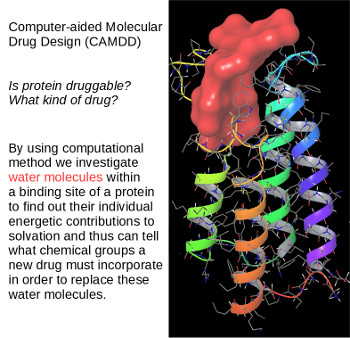
The idea of personalized medicine is very attractive and promising. Considering that each person has slightly different proteins it seems surprising that one drug can work for so many people. My research interest is finding why differences on surfaces of proteins affect their druggability. This is done by testing each water molecule around a binding pocket for its energetic contribution. If water is to be displaced by a drug then that contribution must be compensated by the drug itself. The method used for this investigation is a statistical mechanical framework based on inhomogeneous fluid solvation theory (IFST). Once such contributions are determined one can rationally design molecules containing chemical groups that displace individual water molecules and offer even stronger compensations. The technique used for this part is Computer-aided Molecular Drug Design (CAMDD). The entire invesitgation can be performed for a slightly different protein, which leads to personalized drugs. We use both IFST and CAMDD to speed up the process of drug discovery conducted by our collaborators at the Medical Research Council Cancer Unit.

In Plain English
My interest is in reaction mechanisms of receptors and enzymes in solution. These are large protein structures made of same building blocks (amino acids) having same structural motifs (helices and beta sheets) and both operating on small molecules (hormones or vitamins for example). Yet they differ dramatically in their mechanisms when operating on these molecules. Upon interaction with a small molecule an enzyme will change the molecule while stay unchanged itself but a receptor will change itself while leaving the molecule unchanged. These mechanisms often take place in presence of water molecules. I find out how these water molecules influence the mechanism and I find the way to promote or inhibit the mechanism. Since diseases are result of protein dysfunction then my ultimate goal is to design another small molecule (a drug) to interfere with these mechanisms and help body restore its healthy state.
Featured Publications
- Exploring the role of water in molecular recognition: predicting protein ligandability using a combinatorial search of surface hydration sites Journal of Physics: Condensed Matter 28 344007 2016