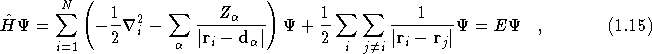
Density Functional theory[7, 8] is a formally exact theory based on the charge density of a system. Kohn-Sham Density Functional theory[9] is a formally exact one-electron theory. Working within the Born-Oppenheimer approximation, the many-body Schrödinger equation,
where ![]() is the many-body wavefunction, is replaced by a
set of N one-electron equations of the form
is the many-body wavefunction, is replaced by a
set of N one-electron equations of the form
![]()
where ![]() is a single-electron wavefunction. These
one-electron equations contain a potential
is a single-electron wavefunction. These
one-electron equations contain a potential ![]() produced by
all the ions and the electrons. Density Functional theory properly
includes all parts of the electron-electron interaction, i.e. the
Hartree potential
produced by
all the ions and the electrons. Density Functional theory properly
includes all parts of the electron-electron interaction, i.e. the
Hartree potential
![]()
where ![]() is the charge density of all the electrons, a potential
due to exchange and correlation effects,
is the charge density of all the electrons, a potential
due to exchange and correlation effects, ![]() , and the
external potential due to the ions,
, and the
external potential due to the ions, ![]() ,
,
![]()
Hohenberg and Kohn[10] originally developed Density Functional theory for application to the ground state of a system of spinless fermions. In such a system the particle density is given by
![]()
with ![]() being the many-body ground state wavefunction of the
system. It can be shown that the total ground state energy of the
system is a functional of the density,
being the many-body ground state wavefunction of the
system. It can be shown that the total ground state energy of the
system is a functional of the density, ![]() , and that if
the energy due to the electron-ion interactions is excluded the
remainder of the energy is a universal functional of the density,
, and that if
the energy due to the electron-ion interactions is excluded the
remainder of the energy is a universal functional of the density,
![]() (i.e.
(i.e. ![]() does not depend on the
potential from the ions). The most elegant proof of Density
Functional theory is due to Levy[11] and is as
follows:
does not depend on the
potential from the ions). The most elegant proof of Density
Functional theory is due to Levy[11] and is as
follows:
For a particular N-representable density![]() (i.e. any density given by an antisymmetric N-electron
wavefunction), a functional of the density corresponding to any
operator
(i.e. any density given by an antisymmetric N-electron
wavefunction), a functional of the density corresponding to any
operator ![]() can be defined via
can be defined via
![]()
The right hand side is evaluated by searching over wavefunctions,
![]() , which give rise to the density
, which give rise to the density ![]() and looking for
the one which makes the expectation value of the operator
and looking for
the one which makes the expectation value of the operator ![]() a
minimum.
a
minimum.
We can define ![]() in the same way, where
in the same way, where
![]()
and
Now let ![]() be the ground state of an N-electron system and
be the ground state of an N-electron system and ![]() a state which yields a density
a state which yields a density ![]() and minimises
and minimises
![]() .
Then, from the definition of
.
Then, from the definition of ![]() ,
,
![]()
Now ![]() is the electronic Hamiltonian, from
Eq.(
is the electronic Hamiltonian, from
Eq.(![]() ), and so
), and so ![]() must obey the
variational principle (see section
must obey the
variational principle (see section ![]() ),
),
Also, from the definition of ![]() , in Eq.(
, in Eq.(![]() ), we
have
), we
have
![]()
since ![]() is just one of the trial wavefunctions
that yield
is just one of the trial wavefunctions
that yield ![]() . Adding
. Adding ![]() to the above equation gives
to the above equation gives
![]()
which in combination with Eq.(![]() ) yields the desired result
) yields the desired result
![]()
hence completing the proof.